Background
The proportion of euploid embryos in IVF PGT (Pre-implantation genetic testing) cycles decreases significantly with advancing maternal age. Recruitment of an adequate number of follicles is crucial in PGT cycles, and it can be regulated by administration of proper gonadotropin doses.
The objective of this study was to compare euploidy and pregnancy rates after administration of different dosages of gonadotropins in PGT cycles.
Materials & methods
A retrospective study of SNP PGT outcome data was conducted to identify differences in euploidy and clinical pregnancy rates. Patients were divided into three gonadotropin dosage groups (<3000 IU, 3000-5000 IU, >5000 IU per IVF cycle) and in four age groups (≤34, 35-37, 38-40, ≥41 y.o.).
406 cycles of IVF treatment with PGT between January 2013 and November 2015 were included in the study (average number of embryos for ET – 1.17). In 132 IVF cycles (109 patients, average age – 35.37±2.21) less than 3000 IU of gonadotropins were administered, in 196 IVF cycles (153 patients, average age – 37.79±3.01) from 3000 to 5000 IU of gonadotropins were administered and in 78 IVF cycles (59 patients, average age – 39.67±4.19) over 5000 IU of gonadotropins were administered.
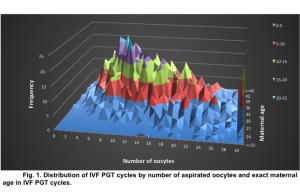
The average maternal age of patients included in the study was 37.49±3.33. The average number of oocytes per retrieval was 16.26±9.05. In 303 out of 406 IVF PGT cycles 10 or more oocytes were aspirated (Fig.1).
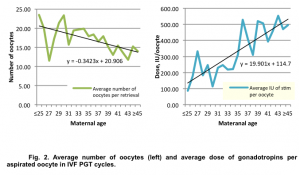
The average number of oocytes aspirated in PGT cycles decreased inconsiderably with advancing maternal age: from 23.5±7.2 in group of patients under 35 y.o. to 14.0±6.11 in group of patients over 45 y.o. At the same time we observed a significant increase in gonadotropin dosage per oocyte retrieved from 85.6 IU/egg in a group of patients under 25 y.o. to 496.4 IU/egg in a group of patients over 45 y.o. (Fig.2).
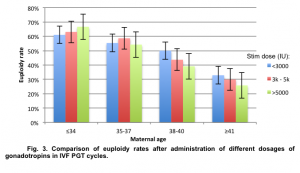
Euploidy rates in the young patient group (≤34 y.o.) were similar regardless of gonadotropin dosage used during controlled ovarian stimulation: 61.03% in the group of patients with low gonadotropin dose (<3000 IU), 63.09% in the group of patients with medium dose (3000-5000 IU), and 66.67% in the group of patients with high gonadotropin dose (>5000 IU). The difference in euploidy rates between the three gonadotropin dosage groups was not statistically significant (p=0.5, χ2=0.454).
In the group of patients 35-37 y.o. euploidy rates ranged from 54.54% to 58.63% (p=0.253, χ2=1.31), in the group of patients 38-40 y.o. euploidy rates ranged from 39.10% to 50.00% (p=0.283, χ2=1.153) and in the group of patients over 40 y.o. euploidy rates ranged from 25.78% to 32.86% (p=0.623, χ2=0.238) (Fig. 3).
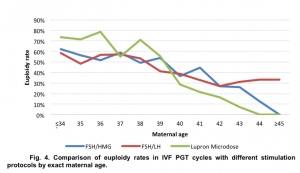
Our data also demonstrated no statistically significant difference in euploidy rates between embryos created in IVF PGT cycles with different stimulation protocols. The association between euploidy rates and stimulation protocols was assessed by identifying coefficients of linear regression: for FSH/HMG stimulation protocol y=-0.0505x+0.7271, for FSH/LH stimulation protocol y=-0.0282x+0.6102, and Lupron Microdose protocol y=-0.0811x+0.9266 (Fig 4).
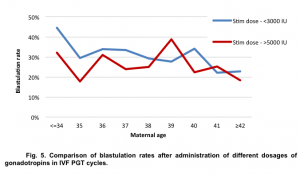
The average blastulation rate (number of embryos available for biopsy divided by number of 2PNs) in PGT cycles where less than 3000 IU of gonadotropins were used for ovarian stimulation was statistically higher than in PGT cycles where over 5000 IU of gonadotropins were used (34.7% and 24.2% respectively, p<0.05, χ2=48.48301). The difference in blastulation rates had a tendency to decrease with advancing maternal age. In a group of patients over 38 y.o. the difference in blastulation rates was not statistically significant (27.0% and 23.5% respectively, p<0.05) (Fig.5).
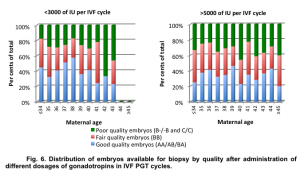
We observed that regardless of total gonadotropin dosage administered in IVF PGT cycle, the proportion of good, fair, and poor quality embryos available for biopsy remained stable, with a slight decrease in the number of good quality embryos with advancing maternal age (Fig.6).
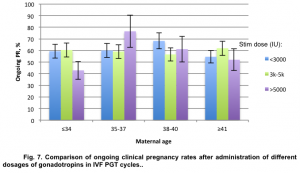
Ongoing pregnancy rates were not statistically different within all study groups (p=0.422, χ2=0.645) and ranged from 42.86% to 60.42% in group of patients under 35 y.o. and from 52.0% to 61.91% in a group of patients over 41 y.o (Fig. 7).
Analysis of ongoing clinical pregnancy rates in different age groups demonstrated no interrelation between maternal age and pregnancy rates after euploid embryo transfer. Using high gonadotropin dosages in a group of patients over 38 y.o. can significantly increase the quantity of aspirated oocytes and at the same time can help maintain proper blastulation, euploidy and pregnancy rates.
Conclusion
Analysis of the data proved that euploidy and ongoing pregnancy rates are not affected by high gonadotropin dosage in IVF PGT cycles. To improve efficiency and cost-effectiveness in IVF PGT cycles the maximum number of follicles recruited in each cycle should be limited only by medical safety concerns and patient well- being.
References