Background
The vast majority of aneuploidies in preimplantation embryos are of maternal origin (either meiotic or mitotic) [1, 2]. The proportion of aneuploid embryos progressively increases with advanced maternal age [3]. Previous studies proved that aCGH and SNP based preimplantation genetic testing (PGT) are the most reliable techniques to determine the genetic status of embryos in vitro [4, 5].
The objective of this study was to identify association between blastocyst morphology and euploidy rates in different age groups in PGT cycles.
Materials & methods
A retrospective comparative study was performed between January 2013 and March 2015. 535 cycles of IVF treatment with PGT were included in the study: in 364 cases blastocysts were analyzed by SNP, and in 171 by aCGH PGT.
A total of 2715 embryos were analyzed for euploidy rates and blastocyst morphology. Morphology of blastocysts was evaluated independently by two embryologists using Gardner classification [6] before trophectoderm biopsy.
Embryos were divided into three groups based on blastocyst quality: good quality (“AA/ AB/BA” – 1057 embryos), fair (“BB” – 962 embryos), and poor (“C-/-C” – 696 embryos) and and into five groups based on maternal age (SART age groups).
Results
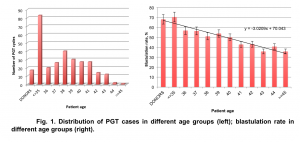
The average maternal age of patients included in the study was 38.03±3.56. 273 cases out of 535 (51%) represent older patient population – 38 y.o and over (Fig.1). The average blastulation rate (number of embryos available for biopsy divided by number of 2PNs) in PGT cycles with donor eggs was 68.08%, while in autologous cycles with PGT it decreased with advancing maternal age from 70.42% (patients under 35 y.o.) to 35.81% (patients over 45 y.o.) (Fig.1).
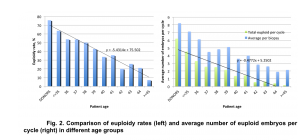
We observed a strong correlation between euploidy rates and patient age. Euploidy rates decrease gradually with advancing maternal age. A decrease in blastulation rate paired with a decrease in euploidy rate caused a rapid reduction in the number of euploid embryos available for embryo transfer in PGT cycles (Fig. 2).
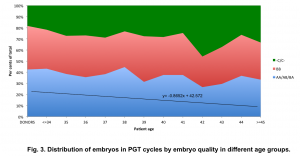
We observed that regardless of patient age, the proportion of good, fair, and poor quality embryos remained stable (Fig. 3), with a slight decrease in the number of good quality embryos (linear regression coefficients:
y = – 0.8652x + 42.572) (Fig. 3).
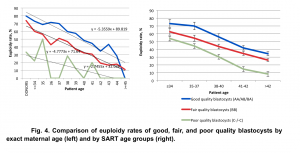
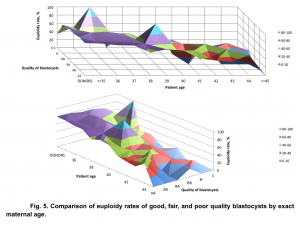
Our data demonstrated a statistically significant difference (p<0.05, χ2=9.72) in euploidy rates between good, fair, and poor quality embryos in all age groups. The biggest difference in euploidy rates between embryos with different morphology was identified among young (<38 y.o.) patients: euploidy rate for good quality embryos was 71.84% (421/586), for fair quality embryos – 58.89% (298/506), and for poor quality embryos -48.87% (173/354) (Fig. 4).
The association between euploidy rates and morphology of the embryos was assessed by identifying coefficients of linear regression: for good quality embryos y=-5.3559x+89.819, for fair quality y=-4.7773x+71.64, and for poor quality embryos y=-2.7455x+32.036.
The difference in euploidy rates between embryos with different morphology has a tendency to decrease with an increase in patient age. For the patients over 40 years old, the euploidy rate for good quality embryos was 38.96% (60/154), for fair quality embryos – 31.25% (50/160) and for poor quality embryos – 11.56% (20/173), p<0.05, χ2=6.08. (Fig. 5).
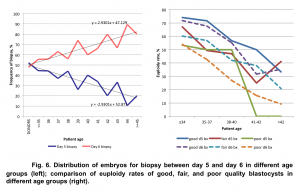
The time embryos became available for biopsy, on day 5 or day 6, reflected dynamics of embryo development in vitro. With advancing maternal age more embryos became available for biopsy only on day 6. The euploidy rate was significantly higher for embryos biopsied on day 5 than on day 6 – 59.62±4.1 and 47.41±3.8, respectively, p<0.05. This pattern was present in all studied groups of embryo morphology (Fig. 6).
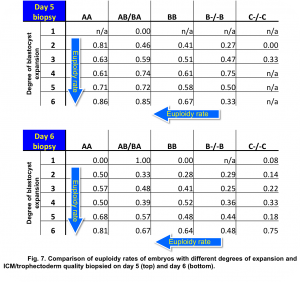
Our data demonstrated a statistically significant difference in euploidy rates between embryos biopsied on day 5 and day 6 with different degrees of blastocyst expansion (Fig. 7).
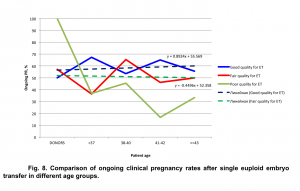
Analysis of the data revealed, that euploid good quality embryos have a higher chance of implantation. Regardless of oocyte source, ongoing pregnancy rates were significantly higher if good quality embryos were available for embryo transfer in PGT cycles (Fig. 8).
Conclusion
Reliable data about the correlation between morphology and euploidy of the embryos will not only help healthcare providers create the best treatment strategy for the infertile couple but will also help patients make an informed decision about the necessity of PGT based on maternal age, quality and quantity of the embryos.